Our lab's research bridges the gap from large-scale genetic discovery to mechanistic understanding. We perform genome sequencing analyses to identify rare genetic variants associated with human disease, applying our methods to both specific disease cohorts and broad "phenome-wide" association studies in datasets like the UK Biobank. This approach has led to the discovery of novel genetic associations for several diseases, including epilepsy, idiopathic pulmonary fibrosis, and diabetes.Realizing the full potential of precision medicine, however, requires translating these genetic discoveries into a functional understanding of disease. To achieve this, we integrate genomics with other large-scale modalities—including transcriptomics, metabolomics, and proteomics. This multi-omic strategy allows us to gain critical insight into the pathophysiology of disease-associated variants. To power future discoveries, we are now leading an initiative to build a large, ancestrally diverse biobank at Texas Children's Hospital, the largest pediatric hospital in the world.

We recently discovered that variants in the gene ITSN1 are associated with both autism spectrum disorder and Parkinson's disease. We now seek to understand its mechanisms and role as a convergent driver of neurodevelopmental and neurodegenerative disease. Our ongoing work includes both mouse and patient-derived iPSC models to dissect how these genetic variants disrupt critical neuronal processes and identify the common molecular pathways that underlie both conditions. We are also partnering up with affected families to understand the phenotypic spectrum of ITSN1-mediated disease (read more).
If you or your loved one has recently been diagnosed and want to learn more or participate in our ongoing research studies, please contact Ryan at ryan.dhindsa [at] bcm.edu.
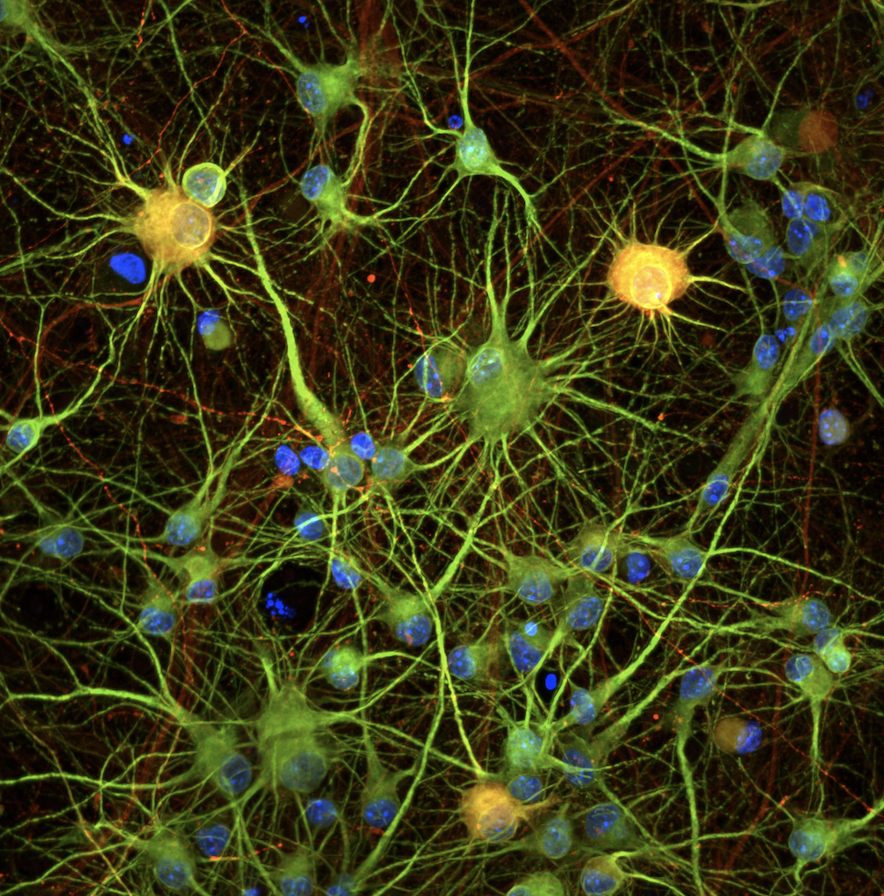
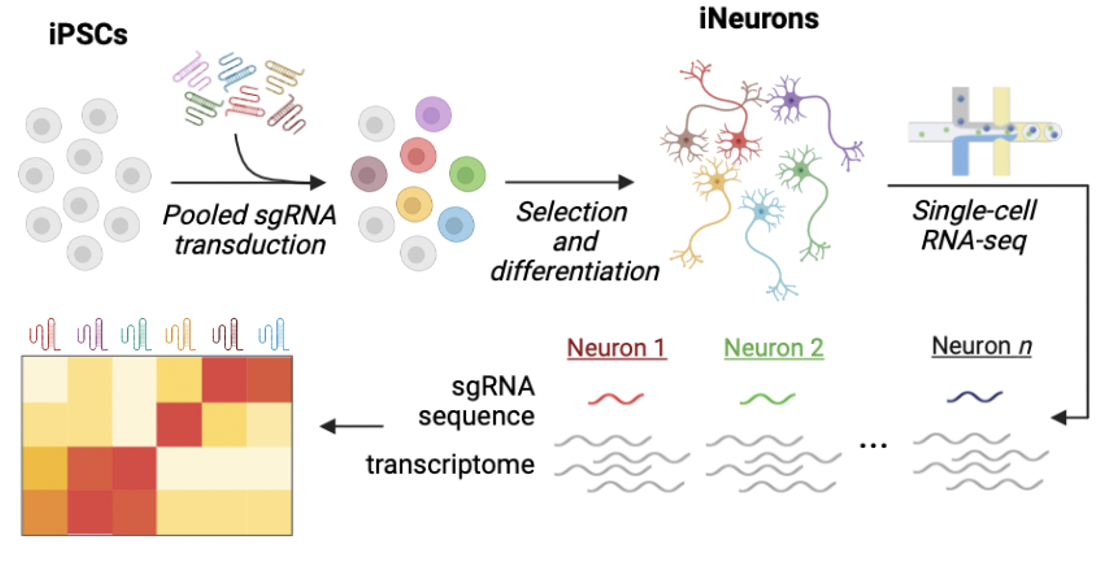
A significant portion (30%) of genes implicated in neurodevelopmental diseases encode transcriptional regulators like chromatin modifiers, transcription factors, and RNA-binding proteins. We hypothesize that many of these distinct genes converge on shared downstream pathways. Identifying these convergent mechanisms is a critical step that could dramatically accelerate drug discovery for intellectual disability, autism spectrum disorder, and epilepsy.To tackle this challenge, our lab utilizes a CRISPR-based functional genomics platform in human iPSC-derived neurons. This system allows us to systematically investigate how disease-causing mutations disrupt neuronal function. We integrate this platform with a variety of advanced functional approaches, including single-cell RNA-sequencing, chromatin profiling, and electrophysiology assays, to map the precise molecular and cellular consequences of these convergent pathways.
Our lab develops new machine learning and AI methods to drive gene and therapeutic discovery. A primary focus is creating computational tools to interpret the flood of data from human genome sequencing. We build robust methods that quantify intolerance to mutation across the genome, allowing us to pinpoint genes and regulatory regions that are critical for human health and are likely sites of disease-causing variants. We have applied these principles to build predictive models that integrate diverse genomic datasets, which we use to nominate novel disease risk genes. We then bridge these genetic insights to therapeutic discovery. By integrating multi-omic data, we sift through complex biological data to identify tractable drug targets and drug repositioning opportunities. This approach allows us to move from a genetic finding to a testable therapeutic hypothesis, accelerating the pace of translational discoveries.
